New CRISPR Method Allows Scientists to Understand Impact of Subtle Mutations
A new method that cleverly uses CRISPR to simultaneously make thousands of point mutations in human genes is helping researchers at Columbia University Vagelos College of Physicians and Surgeons understand how subtle changes to genes contribute to cancer and other diseases.
The method, created by a team led by Alberto Ciccia, PhD, associate professor of genetics and development in the Herbert Irving Comprehensive Cancer Center, was designed to study genes involved in DNA repair but could be applied to the study of any type of gene.
The findings, published in the Feb. 18 issue of Cell, could lead to precision medicines tailored to a patient’s exact genetic abnormality.
Base editors overcome limitations
Researchers trying to understand how a gene impacts disease have generally been limited to making changes that disable an entire gene and its respective protein. “Although this can reveal the gene’s dominant function, it doesn’t tell us about all the other functions it may have,” Ciccia says.
With a new type of gene editing—called CRISPR-dependent cytosine base editing—a single cytosine (C) in a gene can be replaced by a single thymine (T).
“With base editors, we can make subtle changes in any part of the gene, much like what happens in nature,” Ciccia says.
Making the technique even more powerful, the Ciccia laboratory has devised a way to make cytosine base edits across an entire gene and in dozens of genes at the same time—a high-throughput approach that will greatly accelerate the discovery and analysis of clinically important mutations.
New clinically relevant mutations found in DNA damage response genes
Using the new screening method, Ciccia and his team made thousands of mutations in 86 different DNA repair genes and exposed cells with the mutations to DNA-damaging agents to determine if the point mutations altered the function of these genes.
DNA repair genes sense and correct errors in DNA, but mutations within the repair genes can hinder this natural proofreading mechanism. Genetic alterations of DNA repair genes have been implicated in breast and other cancers, along with other human genetic disorders.

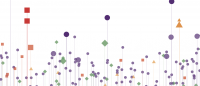
Using base editors to make individual point mutations inside a gene, Ciccia's team can evaluate how each mutation alters cell behavior. In this case, the graphic illustrates how cells carrying a mutation in the ATM gene respond to a drug used to treat advanced ovarian cancer. Image from Cuella-Martin et al. (2021).
The screen successfully identified more than 100 new, likely pathogenic mutations in DNA repair genes, including several in the breast cancer genes, BRCA1 and BRCA2. The full results of the screen are available on the Ciccia laboratory’s website.
“We anticipate this resource will expedite studies of DNA repair genes in human disease,” Ciccia says, “and that base-editing screening technologies will help researchers determine the causes of many different human diseases and accelerate the path to precision medicine.”
References
More information
The study is titled, “Functional interrogation of DNA damage response variants with base editing screens.”
The other contributors are: Raquel Cuella-Martin, Samuel B. Hayward, Xiao Fan, Xiao Chen, Jen-Wei Huang, Angelo Taglialatela, Giuseppe Leuzzi, Junfei Zhao, Raul Rabadan, Chao Lu, and Yufeng Shen (all at Columbia).
This work was supported by grants from the National Institutes of Health (R01CA197774, R01CA227450, and P01CA174653), the EMBO Long-Term Fellowship, the Damon Runyon Cancer Research Foundation, and Pew-Stewart Scholar for Cancer Research program.
Cuella-Martin, Hayward, and Ciccia have filed a provisional patent application derived from this work. The authors declare no other conflicts of interest.