
Living Drugs for the Skin, and Beyond
In the lab, Columbia’s new chair of dermatology, Aimee Payne, is transforming cells into living drugs for autoimmune diseases of the skin
If you want to understand where Aimee Payne, Columbia’s new chair of dermatology, gets her drive and determination, her father would be a good place to start.
Payne’s father was born in North Korea and was conscripted as a teenager into the army during the Korean War. He escaped, only to be shot in the chest and imprisoned by the Americans. In captivity, he taught himself English (one of seven languages he would eventually master), a skill he parlayed into a job as a military interpreter. After earning a scholarship to attend college in the United States, he married, started a family, and eventually settled in Columbus, Ohio, where he began a long career in information technology.
“For my father, Columbus was heaven on earth, and we rarely traveled far,” Payne recalls. “Until I went to college at Stanford, I had never even been on an airplane. So, I wanted to go as far away as possible. California sounded exciting and new.”
Initially planning to be an economics major, Payne dreamed of opening a chain of luxury hotels—until a family friend, a biophysics professor, opened her eyes to the wonders of science. She switched to biology and found a mentor in a physician-scientist studying DNA repair and skin cancer. Before long, she was in the MD-PhD program at Washington University in St. Louis, graduating in 2001.
Payne chose to specialize in dermatology because “almost every health issue has a link to the skin, so there’s no limit to what you can study—cancer, immunology, aging, neuroscience. And by answering fundamental questions about skin disease, we’re also answering questions about human biology,” Payne says.
A one-and-done autoimmunity treatment?
After a residency and postdoc at the University of Pennsylvania, Payne opened her own lab at the university.
She gravitated to the study of a rare disease called pemphigus in which the immune system mistakenly attacks the skin, leading to painful, blistering sores. The top layer of skin can peel off and, without treatment, pemphigus can be fatal. Current therapies suppress the entire immune system to diminish the attack on the skin, but that leaves patients vulnerable to infectious diseases.
A precision therapy for pemphigus, based on Payne’s work, is now in clinical trials and could be a platform for the treatment of many other autoimmune disorders.
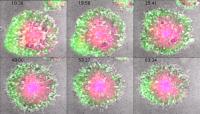
Precision treatment. Still images from a movie of a CAART cell reacting to its target molecule. From Ellebrecht et al. Science (2016) doi: 10.1126/science.aaf6756.
Payne’s treatment is a clever twist on chimeric antigen receptor T-cell (CAR-T) therapy, a form of immunotherapy that harnesses the power of a patient’s own immune system to fight some blood cancers. In CAR-T therapy, T cells are collected from a patient, engineered in the laboratory to recognize and kill the cancer cells, and infused back into the patient.
Following the success of CAR-T therapy in treating blood cancers, researchers began repurposing the therapeutic cells to treat autoimmune diseases and target and destroy the B cells that go rogue in autoimmune disease. Though this approach has shown some promise, the therapy works like a sledgehammer, indiscriminately killing all B cells, not just the smaller number causing the disease.
Payne’s more surgical solution primes T cells to attack only rogue B cells. Payne’s postdoctoral fellowship mentor had discovered that in pemphigus, the rogue B cells target a protein called desmoglein-3, which holds the skin’s epithelial cells together.
A postdoc in Payne’s lab, Christoph Ellebrecht, came up with the initial idea of therapeutic T cells engineered to express desmoglein-3, which causes the cells to selectively stick to, and attack, pemphigus B cells that are covered with desmoglein-3 autoantibodies. Payne calls this approach chimeric autoantibody receptor T-cell (CAART) therapy. Working with Mike Milone, an inventor of the first FDA-approved CAR-T therapy, Ellebrecht and Payne created human CAART cells and tested them in mice, finding that the cells successfully eliminated pemphigus B cells with no apparent signs of toxicity.
CAART cells are designed to go dormant inside the patient after killing pemphigus B cells and could potentially resurface if the B cells come back, potentially keeping the disease at bay for life.
“The beauty of this approach is that it’s a living therapy and could potentially be a one-and-done treatment that puts the disease in remission without causing global immune suppression,” Payne says.
New directions
In 2018, Payne founded a biotech startup, Cabaletta Bio, to refine her approach. “The project reached a point where it was beyond the ability of traditional grants and funding sources to support—the so-called “valley of death” where discoveries from the laboratory often fail to advance,” Payne says. Cabaletta is currently testing the CAART cells in a phase 1 trial for pemphigus patients. It is the first precision cellular immunotherapy for autoimmune disease to enter clinical trials.
The design of CAART therapy also gives it the potential to treat other autoimmune diseases that are caused by a single type of rogue B cell, including some forms of myasthenia gravis, which causes muscle weakness. Cabaletta Bio is currently testing a CAART therapy in patients with muscle-specific tyrosine kinase (MuSK) antibody-positive myasthenia gravis.
“Cell therapies for autoimmunity are now one of the hottest areas for investigation,” Payne says. “With the investments Columbia and NYP are making in cellular therapy, combined with a really exciting cell therapy initiative with the Chan Zuckerberg Institute, we can help move these sorts of therapies from the laboratory to patients in clinical trials.”
Payne joined the Columbia faculty in late 2023, adding the role of department chair to her responsibilities as a lab head and practicing physician, a challenge she seems to take in stride. “The skills you need to run a department are not dissimilar to those you need to navigate your way through your postdoc, negotiate a faculty startup package, get your first NIH R01 grant, and set up and manage a laboratory,” she says.
At the same time, she acknowledges these steps aren’t easy and, as chair, she aims to support young faculty as they grow their careers and nourish more senior faculty who sustain the academic mission. “I was fortunate to have good mentors and role models along the way,” Payne says, “but they’re not always easy to find or available, especially for women and under-represented minorities. It’s hard if you don’t see physicians and scientists that look like you who can help mentor you through the process, so it truly takes a village to make a successful academic clinician-scientist. We have a responsibility to pay it forward to ensure equitable representation in academia.”
References
Aimee Payne, MD, PhD, is the Herbert and Florence Irving Professor and chair of dermatology at the Vagelos College of Physicians and Surgeons and dermatologist-in-chief at NewYork-Presbyterian Hospital.
