Gene Discovery Confirms Role of Serine Deficiency in Rare Eye Disease
Treatments for a rare retinal disease may be on the horizon after a new study has identified gene variants that cause a metabolic deficiency in the eye.
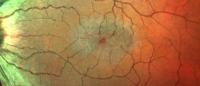
The disease, macular telangiectasia type 2 (MacTel), has been a research focus of Rando Allikmets, PhD, a pioneer in the genetics of eye diseases, for nearly 15 years. MacTel occurs in approximately 1 in 5,000 adults over age 40 and slowly causes a significant loss of central vision, which can impair driving, reading, and other activities.
“MacTel is clearly a genetic disease because it tends to run in families, but it’s been a tough nut to crack,” says Allikmets, the William and Donna Acquavella Professor of Ophthalmic Sciences in the Departments of Ophthalmology and Pathology & Cell Biology at Columbia University Vagelos College of Physicians and Surgeons.
“For many years, we didn’t know what was causing the disease, and we couldn’t find any genes that would give us some clues.”
That began to change a few years ago when Allikmets and an international team of MacTel researchers led by Lowy Medical Research Institute identified two extremely rare mutations that cause the disease.
These genes account for only 0.1% of cases, but their discovery, also made possible by advances in the emerging field of metabolomics, led to a new view of MacTel as a disease of serine deficiency. Serine is one of 20 amino acids that play an important role in metabolism in the eye and throughout the body.
In a landmark study published in NEJM in 2019, the team found that most MacTel patients, regardless of their genetic makeup, have a serine deficiency that leads to the accumulation of a toxic lipid that damages cells in the retina. Some degree of serine deficiency is prevalent in the general population, and some of these individuals can develop various diseases, including MacTel. However, the exact genetic cause is not known for the vast majority of cases.
PHGDH accounts for 3% to 4% of cases
Now the team has uncovered another gene that solidifies the link between MacTel and serine deficiency and points the way to effective treatment. Allikmets, working with David Goldstein, PhD, and the Institute of Genomic Medicine at Columbia, used a recently developed method, called collapsing analysis, that can pinpoint difficult-to-find genes.
The researchers found that mutations in the new MacTel gene, PHGDH, directly reduce serine levels by partially disabling phosphoglycerate dehydrogenase, the most important enzyme involved in serine biosynthesis.
And though PHGDH accounts for only about 3% to 4% of MacTel cases, “its identification is significant because it causes the same disease features that we see in a substantial number of patients with MacTel,” Allikmets says.
“That suggests that fixing the serine deficiency or targeting the toxic lipids may be a way to prevent vision loss in many MacTel patients, regardless of the patient’s precise genetic defect.”
Though the MacTel field is hopeful that treatment options are not far away, Allikmets adds that new therapeutic ideas must be tested first in clinical trials and may not be available for several years.
References
More information
The new study, “Serine biosynthesis defect due to haploinsufficiency of PHGDH causes retinal disease,” was published March 22 in Nature Metabolism.
Authors are Kevin Eade (Lowy Medical Research Institute), Marin L. Gantner (LMRI), Joseph A. Hostyk (Columbia), Takayuki Nagasaki (Columbia), Sarah Giles (LMRI), Regis Fallon (LMRI), Sarah Harkins-Perry (LMRI and Scripps Research Institute), Michelle Baldini (UCSD), Esther W. Lim (UCSD), Lea Scheppke (LMRI), Michael I. Dorrell (LMRI), Carolyn Cai (Columbia), Evan H. Baugh (Columbia), Charles J. Wolock (Columbia), Martina Wallace (UCSD), Rebecca B. Berlow (Scripps), David B. Goldstein (Columbia), Christian M. Metallo (UCSD), Martin Friedlander (LMRI and Scripps), and Rando Allikmets (Columbia).
The research was supported by the Lowy Family; the U.S. National Institutes of Health (grants R01CA234245, R01EY028203, R01EY029315, and P30EY019007); and by funds from Research to Prevent Blindness to the Department of Ophthalmology, Columbia University Vagelos College of Physicians and Surgeons.
The authors declare no competing interests.