A Fasting-Refeeding Paradigm Rejuvenates Old Stem Cells
Feeding after fasting could provide an answer to combatting the effects of old age on the blood stem cells that often lead to life-threatening blood cancer, greater susceptibility to infections, and can even contribute to degenerative disorders in the elderly, according to a study in mice led by researchers in the Columbia Stem Cell Initiative.
As with all stem cells, the body’s blood-forming stems cells become less effective with age or fail to work at all. Blood-forming stem cells not only create red blood cells, but also all the cells of our immune system, and their decline leads to a whole host of problems for the aging body.
The new study indicates that there is a way to turn back the clock to a younger time and rejuvenate these blood stem cells, restoring their ability to regenerate and fight off age-related diseases.
Led by Emmanuelle Passegué, director of the Columbia Stem Cell Initiative, the researchers found that a cellular recycling process known as autophagy must be activated in old blood stem cells for the cells to survive and function in aged animals.
They identify chronic inflammation in the bone marrow microenvironment as the stressor that drives both metabolic impairment and compensatory autophagy activation in old blood stem cells. They further demonstrate that autophagy is an essential survival response in the context of inflammatory signaling and map out the molecular circuitry involved in the autophagy activation process in this context.
And though autophagy helps old blood stem cells survive the stresses of aging, these old stem cells do not have the same capacity as young stem cells to create the blood’s full repertoire of cells. That youthful capacity can be nearly restored, the researchers discovered, with a 24-hour fasting period followed by refeeding.
The findings are important, says Passegué, whose 2017 Nature paper was the first to identify autophagy in aging blood stem cells, because they help scientists better understand what causes stem cell aging and they identify a way to re-awaken the blood-forming function of aged stem cells, which could prevent harmful age-related conditions in the elderly.
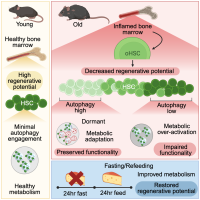
The new study shows that chronic inflammation impairs blood-forming stem cells in old mice, but cells that respond with autophagy can adapt and protect themselves. Feeding after fasting, which triggers transient autophagy, boosts the stem cells' regenerative potential. Figure by Melissa Proven.
Rejuvenation requires resumption of eating
Although previous studies had found that fasting alone can induce autophagy, the new research shows that the resumption of feeding is necessary to fully rejuvenate blood stem cell function. These rejuvenated old stem cells were reset in their metabolism and were almost as proficient as young stem cells at regenerating the blood system upon transplantation and creating new red blood cells, white blood cells, and platelets, which are crucial for preventing anemia and infections.
“Even though the same fasting and refeeding process would be impractical to use directly on people because the equivalent fasting period will be much too long, these findings in mice help us identify the pathways that mimic the effect of fasting and refeeding to rejuvenate old blood stem cells,” Passegué says.
The findings of the new study also provide more evidence to support the idea that fasting can improve cancer therapy by helping blood stem cells enhance a weakened immune system.
Understanding the biological causes of aging stem cells will also provide a better understanding of tissue repair and regeneration, stem cell therapy, and metabolic diseases like coronary artery disease, diabetes, and stroke.
“It doesn’t mean that we will begin living to 200,” says Passegué “But we are in a new frontier, and I think over the next decade we should be able to develop therapeutics that could slow down or stop stem cell deterioration so we can lead healthier lives as we grow old.”
References
More information
Emmanuelle Passegué, PhD, is also the Alumni Professor of Genetics & Development at Columbia University Vagelos College of Physicians and Surgeons.
Co-first authors Paul V. Dellorusso (Columbia) and Melissa A. Proven (Columbia) contributed equally to this work.
Other authors: Fernando J. Calero-Nieto (Cambridge University), Xiaonan Wang (Cambridge), Carl A. Mitchell (Columbia), Felix Hartmann (Stanford University), Meelad Amouzgar (Stanford), Patricia Favaro (Stanford), Andrew DeVilbiss (University of Texas Southwestern Medical Center), James W. Swann (Columbia), Theodore T. Ho (University of California San Francisco), Zhiyu Zhao (UT Southwestern), Sean C. Bendall (Stanford), Sean Morrison (UT Southwestern), and Berthold Göttgens (Cambridge).
The research was supported through grants from the NIH (F31HL151140, TL1DK136048, F31HL160207, R01AG073599, R35HL135763, and P30CA013696), the European Molecular Biology Organization (ALTF-2021-196), Damon Runyon Cancer Research Foundation (DRG-2493-23 postdoctoral fellowships), predoctoral fellowships from the American Heart Association and Hillblom Center for the Biology of Aging predoctoral fellowships, Wellcome (grant 206328/Z/17/Z and core funding to the Cambridge Stem Cell Institute), Cancer Research UK (grant C1163/A21762), Glenn Foundation Research Award, Leukemia & Lymphoma Society Scholar Award, and Milky Way Research Foundation Award (CU21-0225).
Emmanuelle Passegué is a member of the Cell Stem Cell advisory board.
