CRISPR, Stem Cells, and Neonatal Diabetes
Newborns seldom get diabetes, but when they do, it’s almost always caused by a mutation in a single gene that prevents pancreas cells from secreting insulin.
A combination of CRISPR—a gene editing technique—and stem cell technology could replace these defective cells with fully functional insulin-producing cells, Columbia scientists have shown in a new proof-of-principle study. The findings also may help researchers develop stem cell therapies for people with type 1 diabetes, an autoimmune disorder that affects an estimated 1.25 million people in the United States.
The study, published Dec. 11 in Stem Cell Reports, was led by Dieter Egli, PhD, the Maimonides Assistant Professor of Developmental Cell Biology in the Department of Pediatrics at Columbia University Vagelos College of Physicians and Surgeons.
CRISPRed Cells Produce Insulin
To create the cells, the researchers removed a few skin cells from a person with this monogenic form of neonatal diabetes. The researchers converted the skin cells into stem cells and used CRISPR to repair the mutation. They then coaxed the stem cells to create insulin-producing cells.
When transplanted into diabetic mice, these CRISPRed cells produced insulin and were able to control the animals’ blood sugar.

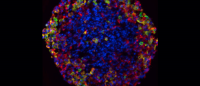
Cells from a patient with neonatal diabetes: uncorrected (left) and cells after CRISPR (right). Images: Dieter Egli/Columbia University Vagelos College of Physicians and Surgeons.
Potential for Treating Neonatal Diabetes
The real test for such cells will come when researchers can transplant them into human patients. That’s still a ways off, since CRISPR can alter other genes in the cell and the transplanted cells can form tumors.
But because the immune system is not involved in monogenic diabetes, unlike type 1, there should be no need to protect the cells from immune attack after the cells are transplanted.
For Egli, that means patients with monogenic diabetes may be the most suitable for initial clinical tests of patient-derived stem cell therapies for diabetes.
“Some forms of monogenic diabetes can look a lot like type 1; both types of patients need to inject insulin to live,” Egli says. “But with such monogenic patients, we probably won’t have to address autoimmunity, and that should make it easier to optimize the cell transplant procedures.”
The CRISPRed stem cells may be a stepping stone to cell transplants for type 1, but the benefit for people with monogenic diabetes shouldn’t be overlooked. “Monogenic diabetes accounts for about 1 to 5 percent of people with diabetes, so it’s not as rare as it seems,” Egli says.
References
More Information
Dieter Egli also is an assistant professor in the Department of Obstetrics & Gynecology at Columbia University Vagelos College of Physicians and Surgeons and a member of the Columbia Stem Cell Initiative and Naomi Berrie Diabetes Center.
The study, “β Cell Replacement after Gene Editing of a Neonatal Diabetes-Causing Mutation at the Insulin Locus,” was published Dec. 11 in Stem Cell Reports.
Other authors: Shuangyu Ma (Columbia University Irving Medical Center), Ryan Viola (CUIMC), Lina Sui (CUIMC), Valentino Cherubini (Salesi Hospital, Ancona, Italy), and Fabrizio Barbetti (Bambino Gesù Children’s Hospital, Rome, Italy).
This work was supported by the American Diabetes Association (grant #1-16-ICTS-029), the Italian Ministry of Health (project PE-2011-02350284), the Russell Berrie Foundation Program in Cellular Therapies of Diabetes, and a scholarship from the China Scholarship Council.
