Building More Homes for Hematopoietic Stem Cells
Study findings could improve stem cell transplantation for the treatment of blood diseases
A new study reveals how stem cell niche generation is regulated in bone marrow, which could lead to improvements in stem cell transplantation for the treatment of blood diseases.
Bone marrow transplants give patients new blood stem cells to replace original diseased cells and have helped millions of people with blood disorders, including cancer.
But for about one in every 10 patients, the introduced cells never take hold in the patient’s bone marrow, leaving patients open to severe infections and bleeding events that are often fatal.
By uncovering the mechanism that creates stem cell niches in bone marrow, the new study from Columbia Stem Cell Initiative researchers could lead to ways to increase the number of niches before transplantation and improve overall engraftment of transplanted cells.
“To improve engraftment, people have generally focused on improving the stem cells themselves, but these stem cells live in niches, specialized microenvironments in the bone marrow that nurture and protect the stem cells,” says Lei Ding, the Gurewitsch and Vidda Foundation Associate Professor of Microbiology & Immunology and Rehabilitation & Regenerative Medicine, who led the research.
“If we can create more niches, even without changing anything else, we should be able to increase the number of transplanted cells that engraft in the bone marrow and reduce graft failure.”
New niche generation requires m6a modifications
Little attention has been paid to how the niche is generated during development, because researchers had largely overlooked this process and considered the niche as a static, pre-existing structure.
To identify mechanisms that control niche development, Ding’s team examined gene activity of mouse bone marrow cells during and after the creation of niches to look for genes upregulated only during niche creation.
This analysis found that genes involved in mRNA processing and RNA methylation, particularly m6a modifications, were highly expressed during niche creation but not after.
That led the researchers to the Mettl3 gene, which is responsible for adding m6a modifications to mRNA, and the discovery that Mettl3 activity is needed to generate stem cell niches. Without Mettl3, Ding’s team found, niche generation was compromised in both number and quality, more bone cells were generated, and fewer hematopoietic stem cells took up residence in the bone marrow.
The researchers also identified a target of Mettl3—Klf2—which must be suppressed by m6a modifications during niche development.
“This study is important because it reveals the first specific mechanism of niche creation,” Ding says, “and shows us that niche creation is controlled genetically.”
“We are super excited about the possibility to create more niches by modulating gene function,” says first author Longfei Gao, a postdoctoral fellow in Ding’s lab.
Artificial niches
The next step for the researchers is to see if they can increase niche generation in adult mice.
“At this stage we’re still doing a lot of genetics to find a driver that can boost niche creation, so we don’t yet have a way to translate our finding to patients,” Ding says.
The findings may also be an important step toward the creation of niche organoids in the laboratory.
“Right now, we can’t culture niche cells very well,” Ding says. “If we can create a niche in the lab, we can better understand how the niche supports stem cells and maybe use such systems to generate more stem cells for patient transplants.”
References
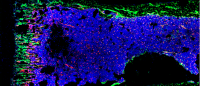
Top image shows normal hematopoietic stem cell niches (pink) in the bone marrow of a mouse. Image provided by Lei Ding.
The research was supported by the National Institutes of Health (grants R01HL153487, R01HL155868, R01GM146061, and P30CA013696), a NYSTEM training grant, an American Heart Association postdoctoral fellowship, a Rita Allen Foundation Scholar Award, a Scholar Award from the Leukemia and Lymphoma Society, and an Irma Hirschl Research Award.
All authors (from Columbia): Longfei Gao, Heather Lee, Joshua H. Goodman, and Lei Ding.
The authors declare no competing interests.
